December 8, 2017: Data and Safety Monitoring in Pragmatic Clinical Trials - Rethinking Clinical Trials

Importance and role of independent data monitoring committees (IDMCs) in oncology clinical trials | BMJ Open
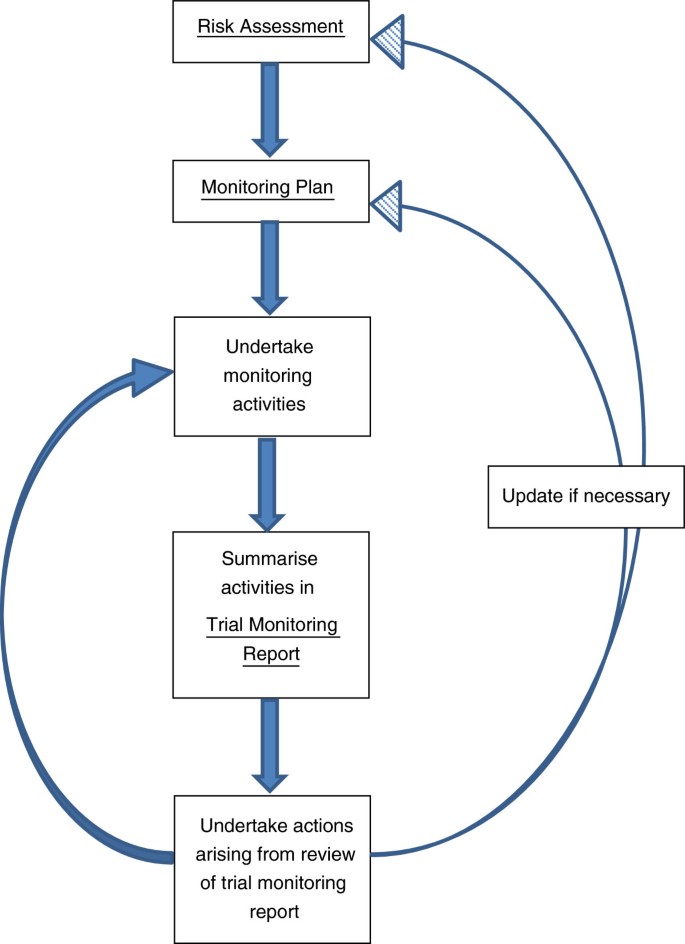
Risk-proportionate clinical trial monitoring: an example approach from a non-commercial trials unit | Trials | Full Text

Data and Safety Monitoring in Clinical Trials Harvey Murff, M.D., M.P.H. GCRC, Vanderbilt University Medical Center. - ppt download
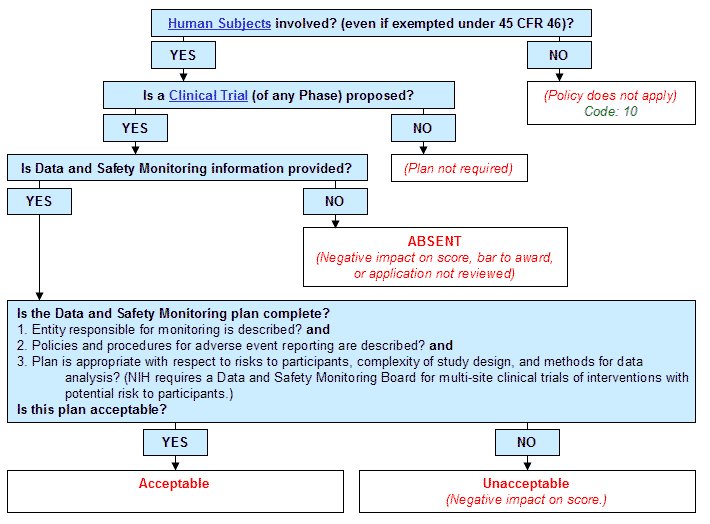
Decision Tree for Data and Safety Monitoring Plan | NIH: National Institute of Allergy and Infectious Diseases

Data Safety and Monitoring Boards Should Be Required for Both Early- and Late-Phase Clinical Trials - ScienceDirect

Data and Safety Monitoring For Clinical Research: Research Ethics: Center for Bioethics: Indiana University


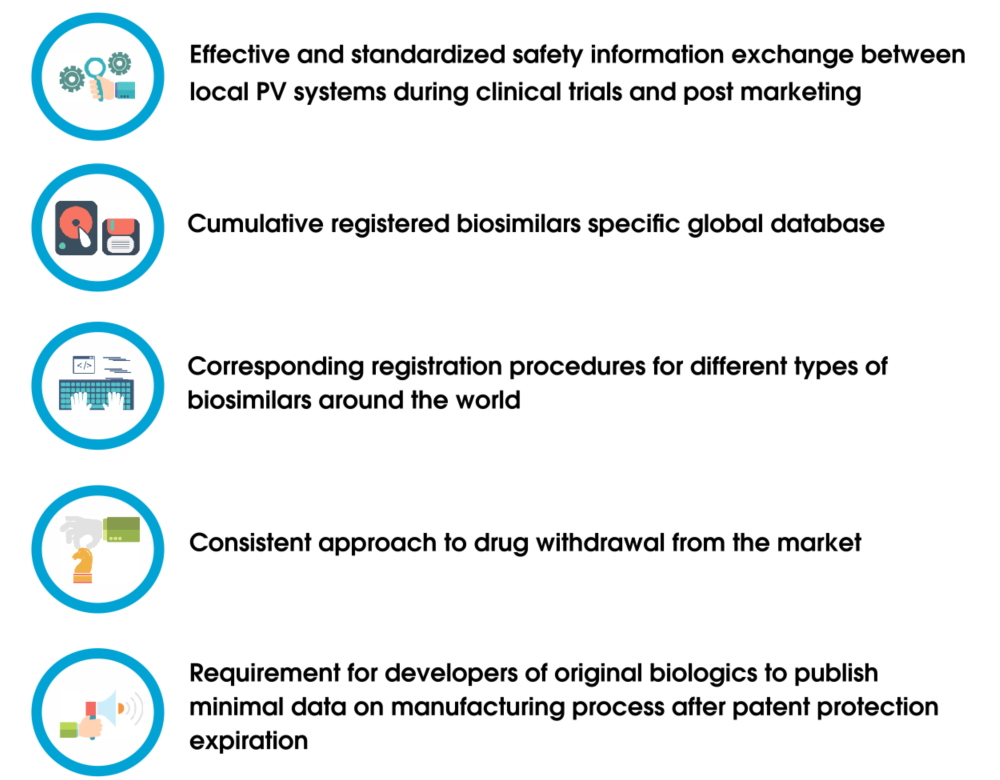




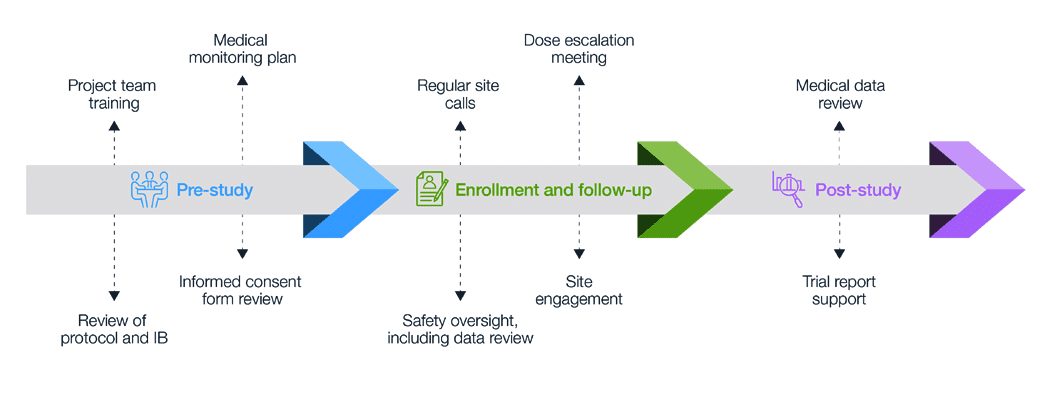



![PDF] Data and Safety Monitoring Board and the Ratio Decidendi of the Trial | Semantic Scholar PDF] Data and Safety Monitoring Board and the Ratio Decidendi of the Trial | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/95d78007a60d58184bdcea808f1f26f279ce7078/23-Table1-1.png)



